3SBio Unveils 2024 Annual Results: Profit Surges 34.9% YoY, Innovation Pipeline Delivers Strong Value
2025-03-25
Chinese leading biopharmaceutical company 3SBio (01530.HK) today announced its 2024 annual results. According to its results announcement, revenue in 2024 grew by 16.5% year on year to approximately RMB 9.108billion. Gross profit added 17.9% year on year to approximately RMB 7.828billion. Net profit attributable to owners of the parent rose 34.9% year on year to approximately RMB 2.09 billion. Net profit attributable to owners of the parent adjusted for non-operating items was approximately RMB 2.318billion, up 18.8% over the same period last year. The board of directors of 3SBio proposed to declare a final dividend of HKD 0.25 per share for the year ended 31 December 2024.
Revenue approaching RMB 10 billion milestone
In 2024, 3SBio achieved strong revenue growth, with total revenue approaching the RMB 10 billion mark. Sales of TPIAO, the Company’s flagship product and the world’s only commercial recombinant human thrombopoietin (rhTPO), totaled RMB 5.062 billion in 2024, reflecting a year-on-year increase of 20.4%. TPIAO has been listed with the highest-level recommendation in several clinical guidelines, including the Chinese Society of Clinical Oncology (CSCO) Clinical Guidelines for the Diagnosis and Treatment of Cancer Therapy Induced Thrombocytopenia (CTIT) (2024 Edition). In 2024, the product received approval for the treatment of primary immune thrombocytopenia in children and adolescents and achieved a renewal in the 2024 National Reimbursement Drug List (NRDL).
EPIAO and SEPO, the recombinant human erythropoietins (rhEPO), maintained their dominant market positions. Yisaipu continued to strengthen its leadership in the domestic market, achieving steady sales growth through proactive market expansion. Cipterbin delivered strong performance, with increasing recognition in clinical applications and longer patient treatment cycles, all of which contributed to rapid sales growth.
In 2024, Mandi, a leading hair loss treatment, achieved sales of RMB 1.337 billion, marking a 18.9% year-on-year increase. The product continued to strengthen its reputation as a science-backed hair growth brand, with its digital marketing system expanding and e-commerce sales rising. Additionally, the approval of Mandi’s foam formulation provided patients with a new treatment option, further solidifying Mandi’s dominant position in the competitive market.
Innovation value steadily coming to fruition
In 2024, 3SBio received approval for the commercial launch of three drugs, including eltrombopag for oral suspension, and submitted new drug applications for four products, including long-acting erythropoietin (SSS06) and anti-IL-7 monoclonal antibody (608). Currently, 3SBio has 30 product candidates within its active pipeline. Among them, there are 13 product candidates in hematology/oncology, 11 product candidates for auto-immune diseases and ophthalmology, 3 product candidates in nephrology and 3 product candidates in dermatology and weight loss. Up to now, 10 candidates have been advanced to the clinical phase III stage.
In the field of nephrology, the Company has established a strong presence across multiple products and indications, including long-acting and small molecules targeting erythropoietin and anemia, enhancing its clinical competitiveness. SSS06 (recombinant erythropoiesis stimulating protein injection) has been approved as an investigational drug for the treatment of chemotherapy-induced anemia (CIA), addressing the gap in long-acting erythropoietin treatment for cancer-related anemia in China. SSS17, the HIF inhibitor with the longest half-life among similar products in development, offers weekly oral administration to significantly improve patient compliance.
The autoimmune disease pipeline is on track to enter a promising phase. In 2024, the phase III clinical trial of anti-IL-17A mAb (608) for the treatment of plaque psoriasis met all efficacy endpoints, and the new drug application has been submitted to and accepted by the National Medical Products Administration. The phase III clinical trials of anti-IL-1β mAb (613) for acute gouty arthritis, and anti-IL-4Rα mAb (611) for atopic dermatitis have all completed patient enrollment. The Company is also advancing new targets and molecules to address unmet clinical needs, aiming to build a competitive and differentiated autoimmune pipeline in China. The investigational new drug applications for anti-BDCA2 mAb (626) for systemic lupus erythematosus (SLE) and cutaneous lupus erythematosus (CLE), in both China and the United States, have been approved, and the phase I clinical trial is ongoing in China. The investigational new drug applications for anti-TL1A mAb (627) for ulcerative colitis (UC) in both China and the United States have been approved. Both candidates are the first investigational drugs approved in China within their respective target categories.
Expanding commercialization landscape through partnerships
In 2024, 3SBio’s external partnerships have delivered strong results. In the consumer health sector, the Company’s subsidiary, 3SBio Mandi, formed a partnership with Shenzhen-based Hybio Pharmaceutical for the weight loss indication of semaglutide injection, leveraging its extensive e-commerce experience to expand patient reach. In oncology, 3SBio reached a partnership with Haihe Biopharma in October 2024 to acquire the commercialization rights for paclitaxel oral solution, Liporaxel. As the world’s only paclitaxel oral solution, Liporaxel has been approved for commercial launch in China. 3SBio will enhance the accessibility of this effective, convenient, and safer chemotherapy drug for more patients through its strong academic capabilities in oncology, pioneering a new model for home-based cancer treatment. Additionally, the Company obtained the commercialization rights for Clifutinib from HEC and for HER2-targeting ADC DB-1303 from Duality Biologics, further diversifying its oncology product matrix.
Moving forward, 3SBio will continue to focus on the fields of its strength, including hematology and oncology, nephrology, autoimmune diseases, hair health and dermatology. The Company will drive value creation for innovative drugs and strengthen the competitive edge of its innovation pipeline. Meanwhile, 3SBio will continue to leverage both in-house R&D and external partnerships, adopting a global perspective to accelerate the development of innovative drugs in a bid to deliver clinical benefits to patients.
Dr. Jing LOU, Chairman and CEO of 3SBio, commented: “2024 marks a year of accelerated progress for 3SBio. We’ve remained committed to advancing innovative drugs, and the results have been both rewarding and widely recognized within the industry. Our core products have continued to post steady sales growth, maintaining leadership positions in their respective categories and demonstrating a robust ability to drive revenue. Clinical trial read-outs for our key pipeline products, 608 and 707, are showing strong promise, paving the way for a bright future. Looking ahead, 3SBio will continue to explore new targets and focus our R&D efforts on areas with significant clinical needs, all while enhancing our innovation pace and capabilities. We will also actively seek partnerships for promising new drugs. Our experience has taught us that while dreams may seem distant, with determination, they are achievable; and while goals may be challenging, perseverance will make them a reality. At 3SBio, we are dedicated to our responsibility as a national biopharmaceutical leader, giving back to society with high-quality, innovative drugs that contribute to the advancement of human health.”
Cautionary Note and Forward-Looking Statements
This press release contains forward-looking statements, such as those relating to business or products outlook, or Company’s intent, plans, beliefs, expectation and strategies. These forward-looking statements are based on information currently available to the Company and are stated herein on the basis of the outlook at the time of this press release. They are based on certain expectations, assumptions and premises, some of which are subjective or beyond our control. These forward-looking statements may prove to be incorrect or may not be realized in the future. With respect to any new product or new indication, we cannot guarantee that we will be able to successfully develop or eventually launch and market such product or indication. Underlying the forward-looking statements is a large number of risks and uncertainties. Further information regarding such risks and uncertainties may be found in our other public disclosure documents. The scientific information involved may only be preliminary and empirical. Shareholders and potential investors of the Company are advised to exercise caution when dealing in the shares of the Company.
Revenue approaching RMB 10 billion milestone
In 2024, 3SBio achieved strong revenue growth, with total revenue approaching the RMB 10 billion mark. Sales of TPIAO, the Company’s flagship product and the world’s only commercial recombinant human thrombopoietin (rhTPO), totaled RMB 5.062 billion in 2024, reflecting a year-on-year increase of 20.4%. TPIAO has been listed with the highest-level recommendation in several clinical guidelines, including the Chinese Society of Clinical Oncology (CSCO) Clinical Guidelines for the Diagnosis and Treatment of Cancer Therapy Induced Thrombocytopenia (CTIT) (2024 Edition). In 2024, the product received approval for the treatment of primary immune thrombocytopenia in children and adolescents and achieved a renewal in the 2024 National Reimbursement Drug List (NRDL).
EPIAO and SEPO, the recombinant human erythropoietins (rhEPO), maintained their dominant market positions. Yisaipu continued to strengthen its leadership in the domestic market, achieving steady sales growth through proactive market expansion. Cipterbin delivered strong performance, with increasing recognition in clinical applications and longer patient treatment cycles, all of which contributed to rapid sales growth.
In 2024, Mandi, a leading hair loss treatment, achieved sales of RMB 1.337 billion, marking a 18.9% year-on-year increase. The product continued to strengthen its reputation as a science-backed hair growth brand, with its digital marketing system expanding and e-commerce sales rising. Additionally, the approval of Mandi’s foam formulation provided patients with a new treatment option, further solidifying Mandi’s dominant position in the competitive market.
Innovation value steadily coming to fruition
In 2024, 3SBio received approval for the commercial launch of three drugs, including eltrombopag for oral suspension, and submitted new drug applications for four products, including long-acting erythropoietin (SSS06) and anti-IL-7 monoclonal antibody (608). Currently, 3SBio has 30 product candidates within its active pipeline. Among them, there are 13 product candidates in hematology/oncology, 11 product candidates for auto-immune diseases and ophthalmology, 3 product candidates in nephrology and 3 product candidates in dermatology and weight loss. Up to now, 10 candidates have been advanced to the clinical phase III stage.
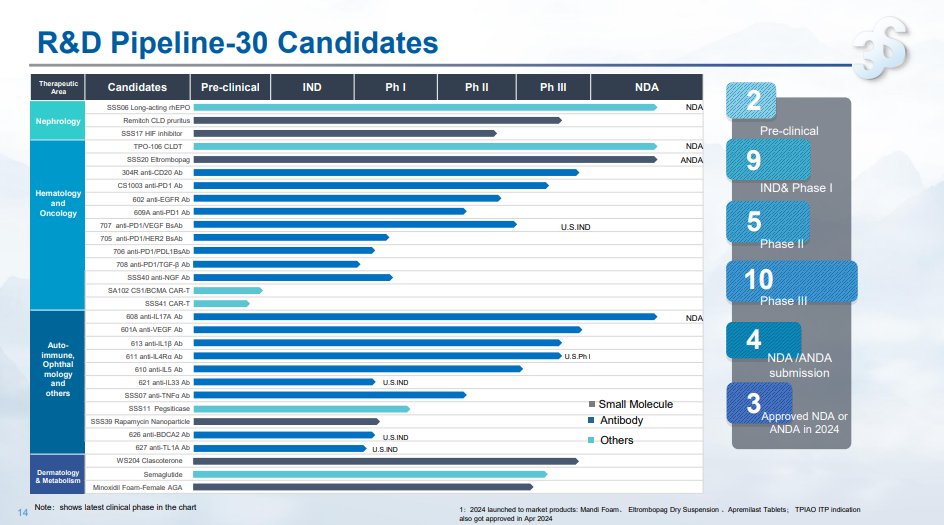
In the field of nephrology, the Company has established a strong presence across multiple products and indications, including long-acting and small molecules targeting erythropoietin and anemia, enhancing its clinical competitiveness. SSS06 (recombinant erythropoiesis stimulating protein injection) has been approved as an investigational drug for the treatment of chemotherapy-induced anemia (CIA), addressing the gap in long-acting erythropoietin treatment for cancer-related anemia in China. SSS17, the HIF inhibitor with the longest half-life among similar products in development, offers weekly oral administration to significantly improve patient compliance.
The autoimmune disease pipeline is on track to enter a promising phase. In 2024, the phase III clinical trial of anti-IL-17A mAb (608) for the treatment of plaque psoriasis met all efficacy endpoints, and the new drug application has been submitted to and accepted by the National Medical Products Administration. The phase III clinical trials of anti-IL-1β mAb (613) for acute gouty arthritis, and anti-IL-4Rα mAb (611) for atopic dermatitis have all completed patient enrollment. The Company is also advancing new targets and molecules to address unmet clinical needs, aiming to build a competitive and differentiated autoimmune pipeline in China. The investigational new drug applications for anti-BDCA2 mAb (626) for systemic lupus erythematosus (SLE) and cutaneous lupus erythematosus (CLE), in both China and the United States, have been approved, and the phase I clinical trial is ongoing in China. The investigational new drug applications for anti-TL1A mAb (627) for ulcerative colitis (UC) in both China and the United States have been approved. Both candidates are the first investigational drugs approved in China within their respective target categories.
Expanding commercialization landscape through partnerships
In 2024, 3SBio’s external partnerships have delivered strong results. In the consumer health sector, the Company’s subsidiary, 3SBio Mandi, formed a partnership with Shenzhen-based Hybio Pharmaceutical for the weight loss indication of semaglutide injection, leveraging its extensive e-commerce experience to expand patient reach. In oncology, 3SBio reached a partnership with Haihe Biopharma in October 2024 to acquire the commercialization rights for paclitaxel oral solution, Liporaxel. As the world’s only paclitaxel oral solution, Liporaxel has been approved for commercial launch in China. 3SBio will enhance the accessibility of this effective, convenient, and safer chemotherapy drug for more patients through its strong academic capabilities in oncology, pioneering a new model for home-based cancer treatment. Additionally, the Company obtained the commercialization rights for Clifutinib from HEC and for HER2-targeting ADC DB-1303 from Duality Biologics, further diversifying its oncology product matrix.
Moving forward, 3SBio will continue to focus on the fields of its strength, including hematology and oncology, nephrology, autoimmune diseases, hair health and dermatology. The Company will drive value creation for innovative drugs and strengthen the competitive edge of its innovation pipeline. Meanwhile, 3SBio will continue to leverage both in-house R&D and external partnerships, adopting a global perspective to accelerate the development of innovative drugs in a bid to deliver clinical benefits to patients.
Dr. Jing LOU, Chairman and CEO of 3SBio, commented: “2024 marks a year of accelerated progress for 3SBio. We’ve remained committed to advancing innovative drugs, and the results have been both rewarding and widely recognized within the industry. Our core products have continued to post steady sales growth, maintaining leadership positions in their respective categories and demonstrating a robust ability to drive revenue. Clinical trial read-outs for our key pipeline products, 608 and 707, are showing strong promise, paving the way for a bright future. Looking ahead, 3SBio will continue to explore new targets and focus our R&D efforts on areas with significant clinical needs, all while enhancing our innovation pace and capabilities. We will also actively seek partnerships for promising new drugs. Our experience has taught us that while dreams may seem distant, with determination, they are achievable; and while goals may be challenging, perseverance will make them a reality. At 3SBio, we are dedicated to our responsibility as a national biopharmaceutical leader, giving back to society with high-quality, innovative drugs that contribute to the advancement of human health.”
Cautionary Note and Forward-Looking Statements
This press release contains forward-looking statements, such as those relating to business or products outlook, or Company’s intent, plans, beliefs, expectation and strategies. These forward-looking statements are based on information currently available to the Company and are stated herein on the basis of the outlook at the time of this press release. They are based on certain expectations, assumptions and premises, some of which are subjective or beyond our control. These forward-looking statements may prove to be incorrect or may not be realized in the future. With respect to any new product or new indication, we cannot guarantee that we will be able to successfully develop or eventually launch and market such product or indication. Underlying the forward-looking statements is a large number of risks and uncertainties. Further information regarding such risks and uncertainties may be found in our other public disclosure documents. The scientific information involved may only be preliminary and empirical. Shareholders and potential investors of the Company are advised to exercise caution when dealing in the shares of the Company.
Return
Prev page: No data
Next page: 3SBio Unveils 2024 Interim Results: Reve...